
This relatively large amount of energy required to go through a change in phase between solids and liquids is referred to as the “latent heat of fusion.” Not only is this much energy needed to be removed to freeze water, but this much energy is also needed to be absorbed to melt 1 gram of ice. The amount of energy that is removed in this change of phase is no longer 4.18 J/g/☌ but 333 J/g. When water freezes it goes through a phase change, liquid to solid. However, if we want to freeze water by going from 1☌ to 0☌ then we will have to take out more than the normal 4.18 J/g/☌ of specific heat. Conversely, if we want to cool 1 gram of water down 1☌ then we will take out 4.18 joules of energy. Therefore, if we want to heat 1 gram (approximately 1 cc or 1 ml) of water up 1☌ then we have to put in 4.18 joules of energy into the water. Water, the most commonly used sensible heat storage device, has a specific heat of 4.18 Joules per gram per degree Celsius (J/g/☌). If a relatively hot object is submerged into a relatively cool body of water then the hot object will cool by giving its thermal energy to the water, and the water will warm.

The most common example is the use of water to cool or heat another substance. In its most basic definition it is, “thermal energy whose transfer to or from a substance results in a change in temperature”. The concept of specific heat, also commonly interchanged with sensible heat, is rather simple for most people to understand.
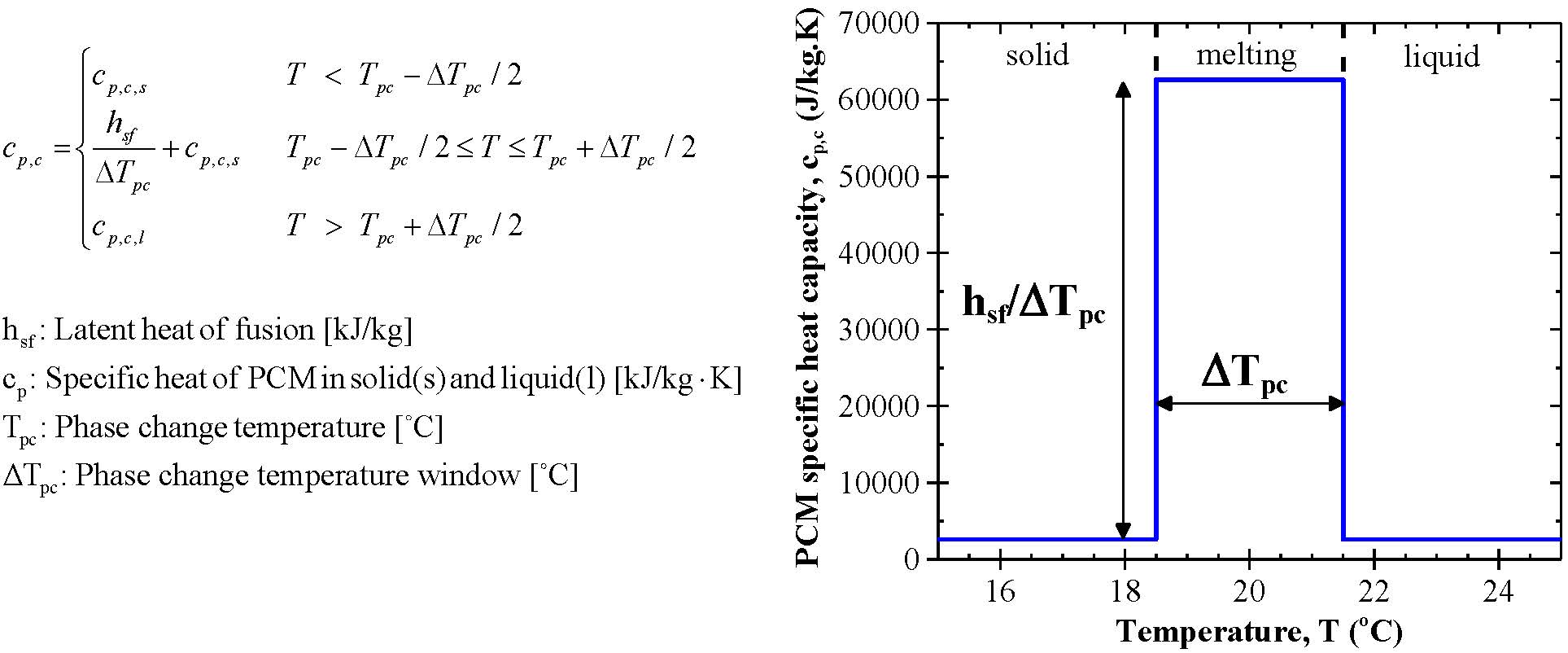
However, a basic understanding of specific heat is needed to fully appreciate latent heat. This paper will primarily focus on the concept of latent heat. These passive thermal energy storage materials can typically be divided into two parts, specific and latent. Passive processes for thermal energy storage have received a lot of attention in the past 25 years. ICNQT researchers work on developing capacity of PCM with nano technology project and find high capacity and eco friendly types of PCM in this project.

